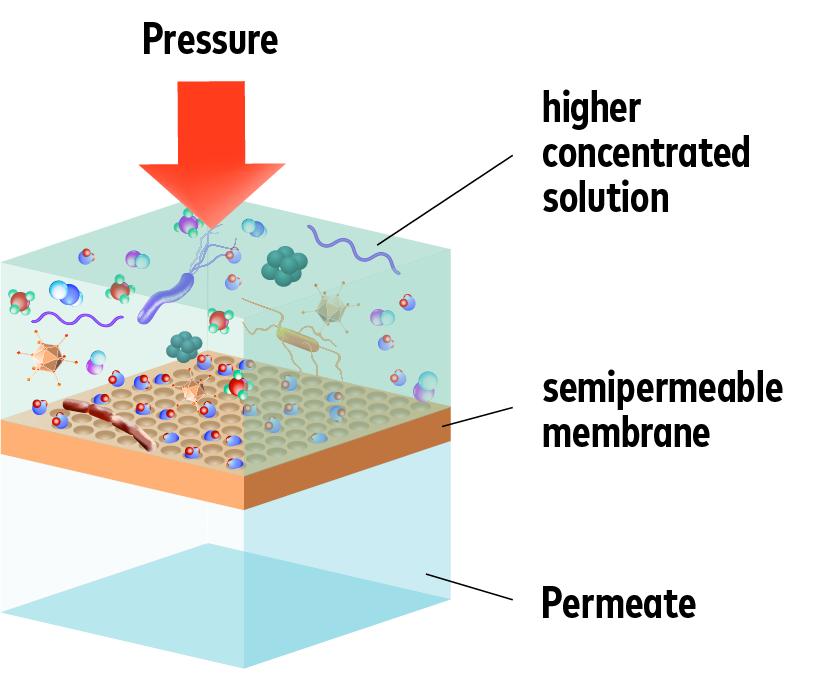
Reverse osmosis is a proven, environmentally friendly and rational way to remove dissolved salts from natural waters. Desalination is carried out by purely physical means – no chemicals are required for the actual process.
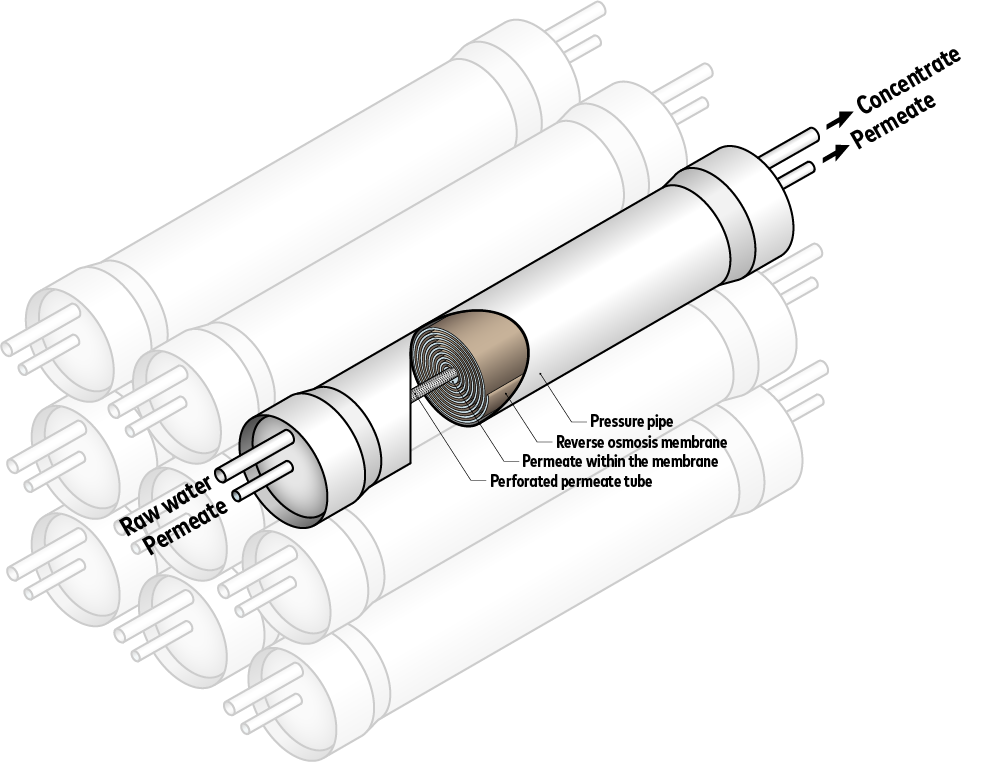
Semi-permeable membranes are used in reverse osmosis. These membranes are permeable for water molecules, but not for the dissolved salts, organic matter, TOC (total organic carbon), bacteria and viruses contained in the water. When the pressure is increased, the water is forced through these membranes – and desalinated.
So much for theory. In practice, the quality of the components and the durability of the membranes used, which have to withstand high pressure loads, are decisive.
How do you recognize a good plant? By the JUDO logo.
JUDO JOS 2 K-S
Ultra-compact reverse osmosis unit
JUDO JOS 2 K
Compact reverse osmosis unit
JUDO JOS 4 - 13 G
Reverse osmosis unit
JUDO JOS 16 - 65 G
Reverse osmosis unit
JUDO JOS 100 - 380 G
Reverse osmosis unit
JUDO CONTIPURE 250 - 1500
Ultrapure water system
JUDO JP 17 - 100 and JEP 17 - 100
Mixed bed multi-way cartridge desalinator
JUDO JROZ 3 - 20
Online reverse osmosis accessories
JUDO JRB 800 - 4000
Permeate collecting tank
JUDO JD 1 SE 3-45, JD 1 CE 3-4 and 5-4
Pressure boosting systems











 © JUDO 2024 | All rights reserved.
© JUDO 2024 | All rights reserved.